DNA analysis of ancient mummy, thought to have smallpox, points to Hepatitis B instead
A team of scientists has sequenced the complete genome of an ancient strain of the Hepatitis B virus (HBV), shedding new light on a pervasive, complex and deadly pathogen that today kills nearly one million people every year.
While little is known about its evolutionary history and origin, the findings confirm the idea that HBV has existed in humans for centuries.
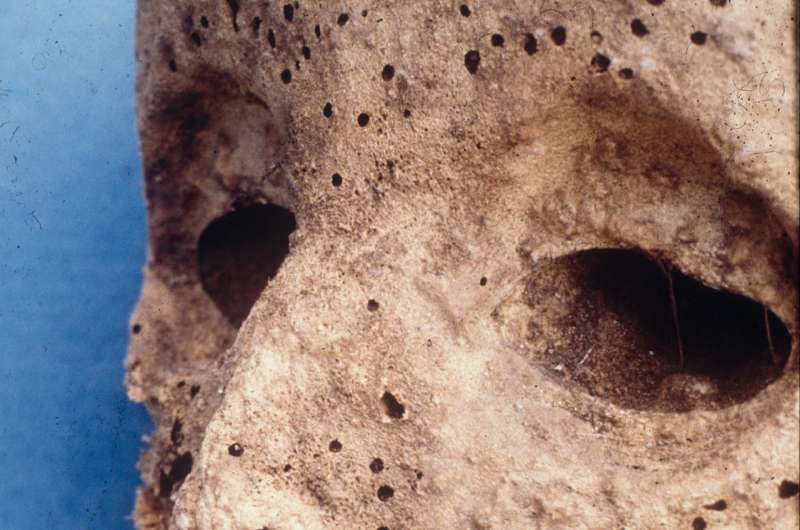
The findings are based on genomic data extracted from the mummified remains of a small child buried in the Basilica of Saint Domenico Maggiore in Naples, Italy.
Previous scientific analysis of the 16th century remains—which did not include DNA testing—suggested the child was infected with Variola virus, or smallpox. In fact, this was the oldest evidence for the presence of smallpox in Medieval remains and a critical time stamp for its origins.
Using advanced sequencing techniques, researchers now suggest otherwise—the child was actually infected by HBV. Interestingly, children infected with HBV infections can develop a facial rash, known as Gianotti-Crosti syndrome. This may have been misidentified as smallpox and illustrates the trickiness of identifying infectious disease in the past.
The findings are published online in the journal PLOS Pathogens.
“These data emphasize the importance of molecular approaches to help identify the presence of key pathogens in the past, enabling us to better constrain the time they may have infected humans,” explains Hendrik Poinar, an evolutionary geneticist with the McMaster Ancient DNA Centre and a principal investigator with the Michael G. DeGroote Institute for Infectious Disease Research.
Using small tissue samples of skin and bone, scientists were able to tease out tiny fragments of DNA and then stitch together pieces of genetic information to create a much more complete picture.
While viruses often evolve very rapidly—sometimes in just days—researchers suggest that this ancient strain of HBV has changed little over the last 450 years and that the evolution of this virus is complex.
While the team found a close relationship between the ancient and modern strains of HBV, both are missing what is known as temporal structure. In other words, there is no measurable rate of evolution throughout the 450-year period which separates the mummy sample from modern samples.
By some estimates, more than 350 million people living today have chronic HBV infections while approximately one-third of the global population has been infected at some point in their lives. Researchers suggest that the underline the importance of studying ancient viruses.
“The more we understand about the behaviour of past pandemics and outbreaks, the greater our understanding of how modern pathogens might work and spread, and this information will ultimately help in their control,” says Poinar.



 Creators of mankind
Creators of mankind Description of “Tall white aliens”
Description of “Tall white aliens” Where they came from?
Where they came from? About hostile civilizations
About hostile civilizations The war for the Earth
The war for the Earth “Tall white aliens” about eternal life
“Tall white aliens” about eternal life Video: “Nordic aliens”
Video: “Nordic aliens” Aliens
Aliens Alien encounters
Alien encounters The aliens base
The aliens base UFO
UFO Technology UFO
Technology UFO Underground civilization
Underground civilization Ancient alien artifacts
Ancient alien artifacts Military and UFO
Military and UFO Mysteries and hypotheses
Mysteries and hypotheses Scientific facts
Scientific facts